The percentage of an element produced in the top producing country. Low = substitution is possible with little or no economic and/or performance impact Medium = substitution is possible but there may be an economic and/or performance impact High = substitution not possible or very difficult. The availability of suitable substitutes for a given commodity. A higher recycling rate may reduce risk to supply. The percentage of a commodity which is recycled. The number of atoms of the element per 1 million atoms of the Earth’s crust. This is calculated by combining the scores for crustal abundance, reserve distribution, production concentration, substitutability, recycling rate and political stability scores. The Chemical Abstracts Service registry number is a unique identifier of a particular chemical, designed to prevent confusion arising from different languages and naming systems.ĭata for this section been provided by the British Geological Survey.Īn integrated supply risk index from 1 (very low risk) to 10 (very high risk). Where more than one isotope exists, the value given is the abundance weighted average.Ītoms of the same element with different numbers of neutrons. This is approximately the sum of the number of protons and neutrons in the nucleus. The mass of an atom relative to that of carbon-12. The transition of a substance directly from the solid to the gas phase without passing through a liquid phase.ĭensity is the mass of a substance that would fill 1 cm 3 at room temperature. The temperature at which the liquid–gas phase change occurs. The temperature at which the solid–liquid phase change occurs. The arrangements of electrons above the last (closed shell) noble gas. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. We offer tutoring programs for students in K-12, AP classes, and college.A vertical column in the periodic table. SchoolTutoring Academy is the premier educational services company for K-12 and college students. Interested in science tutoring services? Learn more about how we are assisting thousands of students each academic year. Silicon and boron compounds are also used in fireworks, as they are less toxic than some other compounds. Many compounds are used to form glassware, especially in chemical and industrial uses, such as optical fibers. Compounds of boron are used as catalysts in many chemical reactions. However, other compounds can be used as disinfectants and antiviral agents. Many compounds of metalloids are highly toxic, such as those containing arsenic and antimony. Germanium is alloyed with silver to make tarnish-resistant sterling silver. For example, boron is used in alloys with steel and with nickel for welding components. The metalloids are often too brittle to be used as pure substances, but form many useful alloys. Silicon and germanium revolutionized the electronics and computer industries. The metalloids, especially silicon, boron, germanium, and compounds of arsenic and antimony, are natural semiconductors. Semiconductor chips, transistors, and other electronic parts form integrated circuits for everything from computers to cell phones. Semiconductors can conduct electricity under some conditions, so electrical current can be controlled. 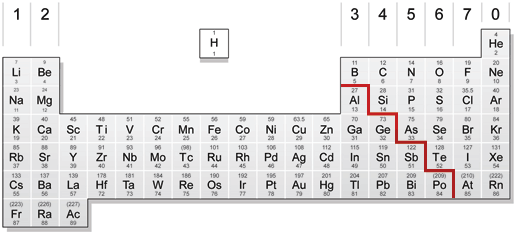
Metalloids are good semiconductors, which mean that they are between the electrical conductivity of metals and materials used for insulation. For example, polonium (Po), and astatine (At) are sometimes included in the list of metalloids, because of their chemical properties and their location on the periodic table. Whether elements are classified as metalloids or not depend upon the chemist’s decision. 
In chemical reactions, they often act more like nonmetals, but they form alloys like metals. Most metalloids are brittle (a non-metallic property), act as semiconductors of electricity, and have a metallic luster (also a metallic property). MetalloidsĮlements are commonly classified as either metals, nonmetals, or metalloids. The most commonly recognized metalloids include the elements boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb), and tellurium (Te). They are on the periodic table along the dividing line between metals and nonmetals. Metalloids are elements that have some properties of metals and some of nonmetals. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
